The use and cultivation of yeast are possibly one of the oldest forms of biotechnology known to humanity. People have been leveraging its natural properties to make products such as bread and beer for generations. In the modern era, scientists have also found that yeasts can be engineered to produce drugs, using chemicals and other industrially important molecules.
Yeast in Human Industry
Yeast is, in fact, a kind of single-celled organism similar to bacteria in evolutionary terms. Saccharomyces cerevisiae is possibly its best-known and widely-used species in the industry today. In fact, many companies may genetically engineer their own variant for greater efficiency or individuality in what the yeast produces (e.g., “S. carlsbergensis,” also known as S. pastorianus). This yeast essentially functions to break down glucose into other forms.
Humans have found that some of these metabolic products like ethanol were useful to them, and thus developed techniques such as fermentation. Today, billions of dollars’ worth of industry revolve around these increasingly sophisticated processes, and the various valuable molecules they can make.
The production of shikimic acid is a prime example of the state-of-the-art yeast-based bioindustry. This molecule is made as a result of an essential biological pathway. The end-point of this process also points to many others. This is due to the structure of shikimic acid, which contains a six-carbon ring of carbon, to which a carboxyl and various hydroxyl groups are attached. Accordingly, the molecule (also known as shikimate) can go on to be converted into many other important biomolecules, which include the aromatic amino acids such as tyrosine, phenylaniline, and tryptophan.
Therefore, shikimate can be considered to be the basic precursor of many other, more complex, biomolecules. They could include the neurotransmitters, serotonin and dopamine, and all their various derivatives and alternate forms. In addition, shikimate could also be converted into numerous valuable industrial products, including succinyl benzoate, coumaric acid, and cinnamic acid. Shikimate is also used to produce the drug oseltamivir (Tamiflu). Therefore, the molecule is an important component in many modern-day industrial and pharmaceutical processes.
Shikimate Production: A Bio-Goldmine
Shikimic acid was named as it was initially discovered in the Japanese flower, shikimi. However, it became apparent that this product was produced in cells across the evolutionary spectrum, from star anise to humans to yeast.
The acid is produced as part of the overall metabolic process that derives energy from glucose and is converted from the common sugar by-products, phosphoenolpyruvate (PEP) and erythrose-4-phosphate (Ery-4-P) into the precursor DAHP, and then into shikimate. This is facilitated by the energy-exchange molecule, NAD.

Shikimic acid has also been sourced from Chinese star anise in the past. (Source: Wikimedia Commons)
Shikimate was traditionally sourced from plants; however, this became economically unfeasible, so techniques revolving around yeast geared to the molecule’s production were developed and implemented. These yeasts greatly improved the efficiency of shikimate bio-manufacturing. However, this form of biosynthesis is also well-regulated in nature and still results in the loss of some resources (mostly metabolic energy and carbon), in non-bioengineered organisms. In addition, some yeast species may not activate the PEP/Ery-4-P conversion pathway, without some kind of external trigger (e.g., light or electricity).
Therefore, there has been room for improvement in the yeast ‘biofactories’ that are steadily taking over the industrial production of shikimate.
A group of scientists from the Wyss Institute for Biologically Inspired Engineering and John A. Paulson School of Engineering and Applied Sciences (SEAS) at Harvard believed that they had a way to address these issues about shikimate.
These researchers, led by Neel Joshi, Junling Guo, and Miguel Suástegu, believed that the emerging technique of bio-hybridization could result in improved shikimate yields from their test yeast. It involved the augmentation of single-celled organisms with nanoscopic semiconductor particles that could convert the light that hits them into extra metabolic energy for the cell in question.
Nano-Electro-Hybrid Yeast
This technique, however exciting in its potential, still has some early teething problems. The main issue is that the semiconductor materials used are often toxic to the cells on which they are placed.
The Wyss/SEAS team got around this challenge by coating their particles (made of indium phosphide) with a sticky polyphenol-based compound. This allowed the particles to be attached to the surface of yeast cells while insulating the said cells from any toxic effects.
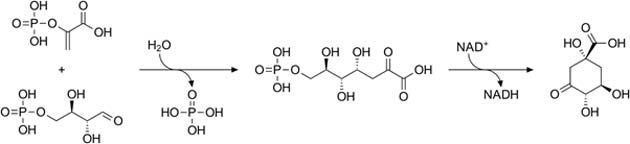
The basic shikimate production pathway. (Source: Wikimedia Commons)
The new nano-semiconductors demonstrated the ability to shuttle additional electrons to the NAD product, NADH. This modification to the overall metabolic system in the yeast led to more energy for more shikimic acid production.
The team reported that their intervention resulted in an 11-fold increase in this process in response to light exposure in the yeast.
Dr. Guo now believes that this new technique could be optimized in terms of both the semiconductor and engineered-yeast components of the bio-hybrids in the future.
This breakthrough is to be published in an upcoming issue in Science journal. It is an exciting first step in the kind of science that melds nano-electronics and biology in new and increasingly effective ways. Who knows, perhaps the chemical and biological technology of the world may be 100% bio-hybridized, one day?
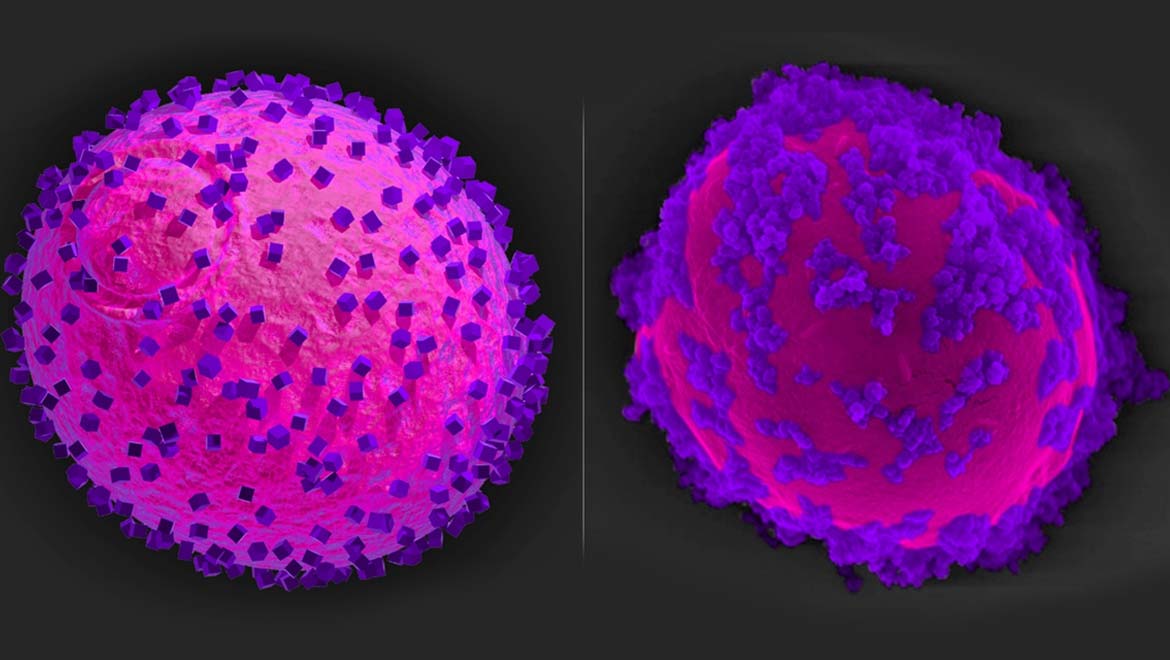
Top Image: The yeast cells (magenta) coated with the nano-semiconductors (purple). Left: researcher’s model illustration; right: actual micrograph of the live yeast. (Source: Wyss Institute at Harvard University)






No comment