Worldwide, heart transplants provide a vital lifeline to over 4000 patients per year. Whilst for those that are lucky enough to receive a donor organ, this can mean both a huge relief a new lease of life, the process of waiting for a suitable organ can be traumatic and worrying.
Unfortunately, the lack of donor organs also means that many people face the worry of a long waiting list, or the prospect of a suitable organ never becoming available. In the US, there are currently around 3000 patients on the waiting list for a heart transplant however, 1000 of these will be disappointed due to the lack of suitable donor organs.
Science is looking for ways to solve this shortage
Science is now looking at ways to regenerate tissues from the small samples which have already been created in the lab, moving towards the potential of creating full-size organs which could, in theory, help us move towards a future where more organ transplants can be carried out. The hope is that the heartache of patients having to miss out on a transplant could be reduced, and the new chance for a healthy life that organ transplants provide could be offered to more people.
The problem, so far, has been that scientists simply haven’t been able to find a way to recreate the network of tiny blood vessels which are needed in order to carry essential nutrients and oxygen, without which tissue growth cannot continue. 3D printing has already been used to create an artificial version of human tissue, but so far researchers have not been able to recreate the tiny networks of blood vessels known as capillaries which are a vital part of our vascular systems.
A team from Worcester Polytechnic Institute (WPI), the University of Wisconsin—Madison and Arkansas State University-Jonesbro have turned to the plant kingdom to try and find a solution to this problem.
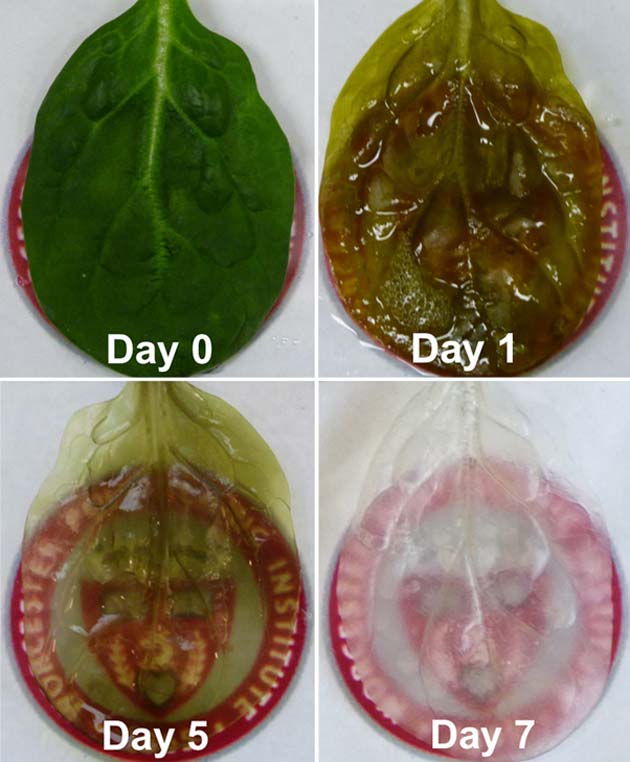
A time lapse of a spinach leaf's transformation. Credit: Worcester Polytechnic Institute
Whilst animals and plants employ very different methods of transporting fluids and other essential molecules, the vascular networks of both plants and animals are surprisingly similar. If you hold the majority of leaves up to the light, you will see a beautiful and detailed network of tiny veins, which both support the leaf and transport carbohydrates and water to the very edges of the structure.
How did the team achieve this impressive goal?
First, the spinach leaves were perfused with a detergent which removed all the cellular material, essentially leaving a scaffolding type structure make of cellulose, which acts to keep the plant cells in place. Joshua Gerslak, a graduate student working within the lab, was responsible for this stage of the process and employed techniques which has previously been used to decellularize human hearts. After this process, the leaf now looks nearly completely transparent and is ready for the next stage.
This part of the process involved placing, or ‘seeding’, cardiac muscle cells onto the initial scaffolding structure. The team, led by Glenn Gaudette from the Myocardial Regeneration Lab of WPI, also lined the veins within the spinach leaf with human cells. Next, a simulation of human blood was created using microbeads of a similar size to blood cells, along with fluids to replicate plasma.
One question that the team wanted to find the answer to was whether or not that once the cardiac cells were attached to the leaf scaffold, would they continue to contract? Amazingly, they did, and to all intents and purposes looked and acted like normal cardiac cells.
Essentially, each leaf provides a thin, flat tissue with a vascular network. The intention is that the leaves can then be ‘stacked’ together in order to create cardiac tissue.
This could help shape the future of transplants
This particular project employed scientists from a wide variety of disciplines which allowed for some fresh perspectives and solutions. The team included human stem cell researchers, biomedical engineers, plant biologists and biotechnologists. As Gaudette said “this project speaks to the importance of interdisciplinary research. When you have people with different expertise coming at a problem from different perspectives, novel solutions can emerge.”
Exciting advances in this field include exploring the possibility of utilizing different plant structures for a variety of different needs. For instance, wood, with its high strength and regular cell structure, could be ideally suited to bone grafts. Plants such as jewelweed, which has hollow cylinders within the stem, just might be a perfect match for replacing or repairing sections of strong artery walls.
A huge advantage of this technique is also that a wide range of plants can be easily cultivated in a controlled environment, allowing for a potentially very reliable source of base material.
The research has so far been published online and will also feature in the May 2017 issue of Biomaterials.
Top image: Human Heart Tissue With the Help of A Spinach Leaf. (wpi.edu)
References
http://www.clevelandclinicmeded.com/medicalpubs/diseasemanagement/cardiology/heart-transplantation/
https://www.sciencedaily.com/releases/2017/03/170322152753.htm
https://www.wpi.edu/news/wpi-team-grows-heart-tissue-spinach-leaves
http://news.nationalgeographic.com/2017/03/human-heart-spinach-leaf-medicine-science/






No comment