Diabetic neuropathy (DR), i.e., the damage of nerves incurred by diabetes, affects more than 200,000 people, and at least half the population of diabetics in the United States. The condition is known to be common among long-time sufferers of diabetes. People living with diabetes could also face the issue of blindness or loss of vision, and DR is the primary cause of these two conditions in the U.S. Now, although diabetic neuropathy cannot be cured, when diagnosed early, it can alleviate certain symptoms and improve quality of life.
Following a clinical trial, in April 2018, the Food and Drug Administration (FDA) cleared for marketing a system called IDx-DR (created by the company, IDx LLC), which uses AI software to detect diabetic retinopathy in adults with diabetes. This is the first time the FDA has approved such an approach. The medical device can reportedly be used by patients and any healthcare providers, even non-eye care professionals, to interpret results, without the help of clinicians.
At the time of the online release, Malvina Eydelman of the Division of Ophthalmic, and Ear, Nose and Throat Devices at the FDA's Center for Devices and Radiological Health, said, “Today's decision permits the marketing of a novel artificial intelligence technology that can be used in a primary care doctor's office.”
AI Diagnostic System, IDx-DR
Details of the IDx-DR system have been published by the developers in an August 2018 issue of Nature Digital Medicine journal. The paper also describes the technology used and the results of the study.
The researchers of the study, led by Michael Abràmoff, determined that AI-based algorithms and machine learning greatly improved diagnostic accuracy. Thus, they created this practice, not only to achieve efficiency in diagnosis, but also to ensure autonomy, or the requirement for review by experts, of its implementation in primary care settings. The aim was also to get a quick yet high-quality image feedback. In addition, importance was given to achieving diagnostic accuracy across race, ethnicity, and age.
For the investigation, 900 adults with diabetes were enrolled across ten primary care centers in the United States. These participants had no known history of diabetic retinopathy. The IDx system procured retinal images of patients with the help of a robotic camera (Topcon NW400), following which the AI analyzed and, accordingly, made clinical diagnoses. The process reportedly took just 20 seconds. This is typically followed by a physician uploading the respective retina-images to a cloud server, using the IDx-DR software.
How did artificial intelligence detect disease? With the assistance of detectors that could identify lesions, including microaneurysms and hemorrhages, related to DR.
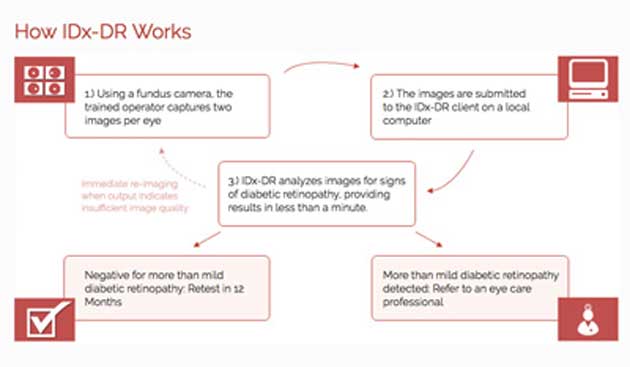
A flowchart demonstrating the steps involved in the diagnosis of diabetic retinopathy via AI diagnostic tool, IDx-DR. (Source: Vision Monday)
Also, for the first time, an AI system was compared with an independent, high-quality gold standard (i.e., certified by the Wisconsin Fundus Photograph Reading Center (FPRC)), which included fundus imaging and OCT (Optical Coherence Tomography) protocols.
Results of the study showed that both methods collected diagnostic data for 819 out of the 900 participants. The FPRC system diagnosed 198 patients with mild diabetic retinopathy and recommended for them further analysis by a specialist. On the other hand, the new AI displayed a sensitivity of 87% and singled out 173 of the 198 participants with the disease. Additionally, this novel method showed a specificity of 90% when it identified 556 of the 621 disease-free subjects. To top it all, the AI had an imageability rate of 96%.
The FDA has made clear that patients who have had eye surgery, laser treatment or any eye injections in the past must not be screened for DR with the AI system. They also urge caution to individuals experiencing prior blurred vision, nonproliferative retinopathy, macular edema, and a list of other previously diagnosed conditions.
Real-World Applications & Future of the AI System
Alvin Liu, a retina specialist and assistant professor of ophthalmology at John’s Hopkins Medicine expressed his enthusiasm for the model. He said, “It's very exciting and it's definitely paradigm shifting. The FDA thinks the results are so impressive, so reliable that whatever the machine says, it does not need a human for verification. The buck stops with the computer.” He added, “It changes the way we think about AI, what it means to be a doctor, and what the future holds.”
In June 2018, a few months following the FDA clearance, Iowa (the Diabetes and Endocrinology Center at the University of Iowa) was the first state in the country to implement IDx-DR to screen diabetic patients for retinopathy.
In the future, as lead author Abràmoff described, this concept could be applied in the early detection of other diseases such as glaucoma and macular degeneration, and it could also take diagnostics like these into primary care settings and retail to further give access to patients.
Currently, the company, IDx LLC, in collaboration with the American Medical Association, is working on the logistics, guidelines, and financials of IDx-DR for providers, suppliers, and physicians.
To sum it up, this latest breakthrough in artificial intelligence and diagnostics, that has beaten all the odds per the clinical trials, could be a benchmark in attaining “high diagnostic accuracy” for patients with diabetic retinopathy; autonomously, at that.
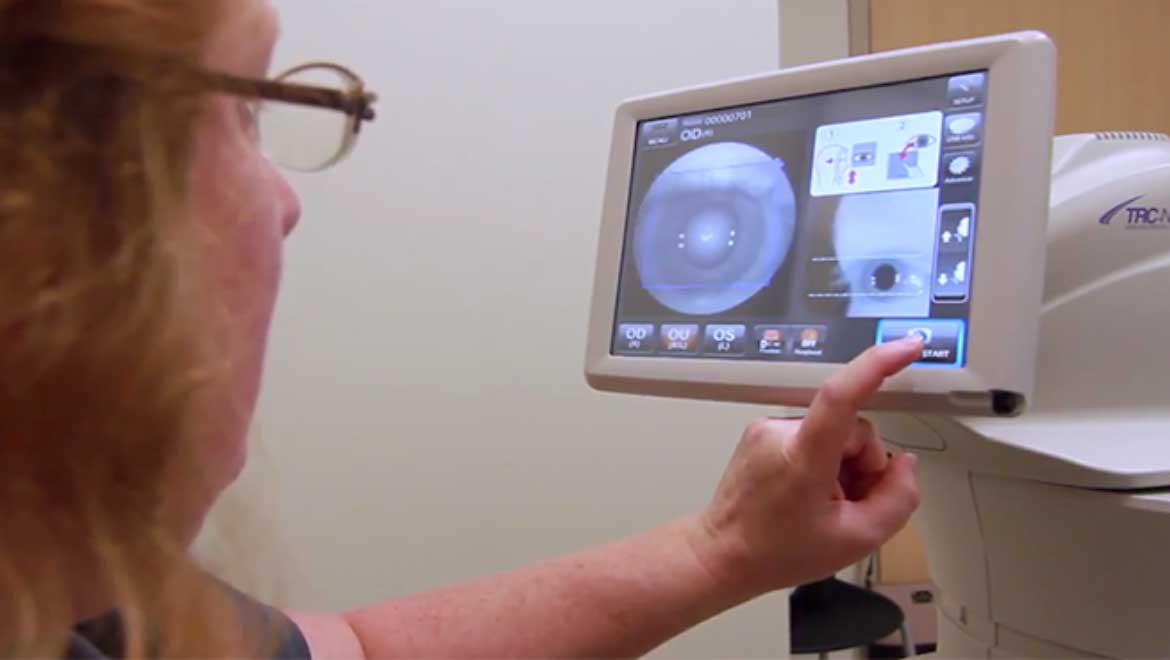
Top Image: IDx-DR, the AI diagnostic system being implemented at a health center at the University of Iowa. (Source: University of Iowa Health Care)
References
[1] Neuropathy (Nerve Damage), American Diabetes Association, http://www.diabetes.org/living-with-diabetes/complications/neuropathy/, (accessed Sept 9, 2018)
[2] FDA OKs AI Device to Detect Diabetic Retinopathy in Primary Care, 2018, Medscape, https://www.medscape.com/viewarticle/895061, (accessed Sept 9, 2018)
[3] Abràmoff, M. D. et al. (2018), Pivotal trial of an autonomous AI-based diagnostic system for detection of diabetic retinopathy in primary care offices, NPJ Digital Medicine, 1 (39)
[4] Artificial intelligence can deliver specialty-level diagnosis in primary care setting, study shows, 2018, ScienceDaily, https://www.sciencedaily.com/releases/2018/08/180828085908.htm, (accessed Sept 10, 2018)
[5] The Numbers Behind the First FDA-Approved Autonomous AI Diagnostic System, 2018, Healthcare Analytics News, https://www.hcanews.com/news/the-numbers-behind-the-first-fdaapproved-autonomous-ai-diagnostic-system, (accessed Sept 10, 2018)






No comment